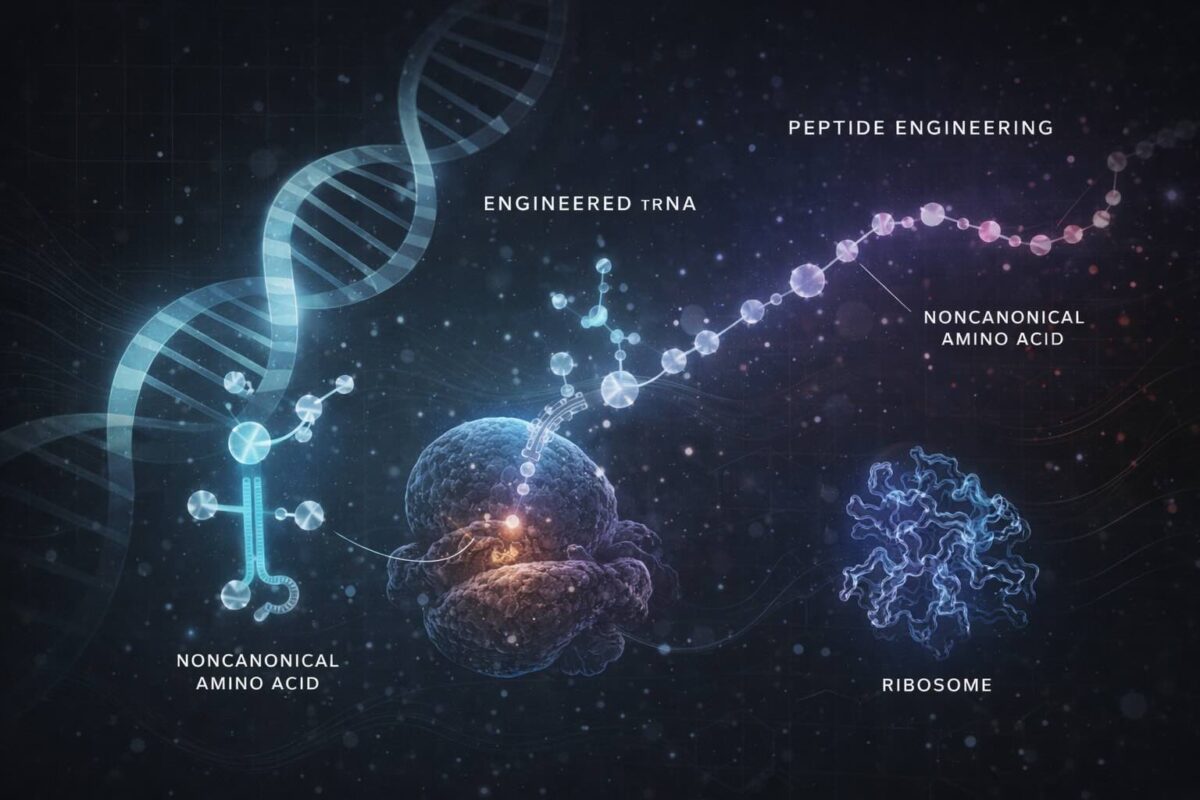
Expanding the Horizon of Peptide Therapeutics: How Engineered tRNAs Enable Ribosomal Incorporation of Noncanonical Amino Acids
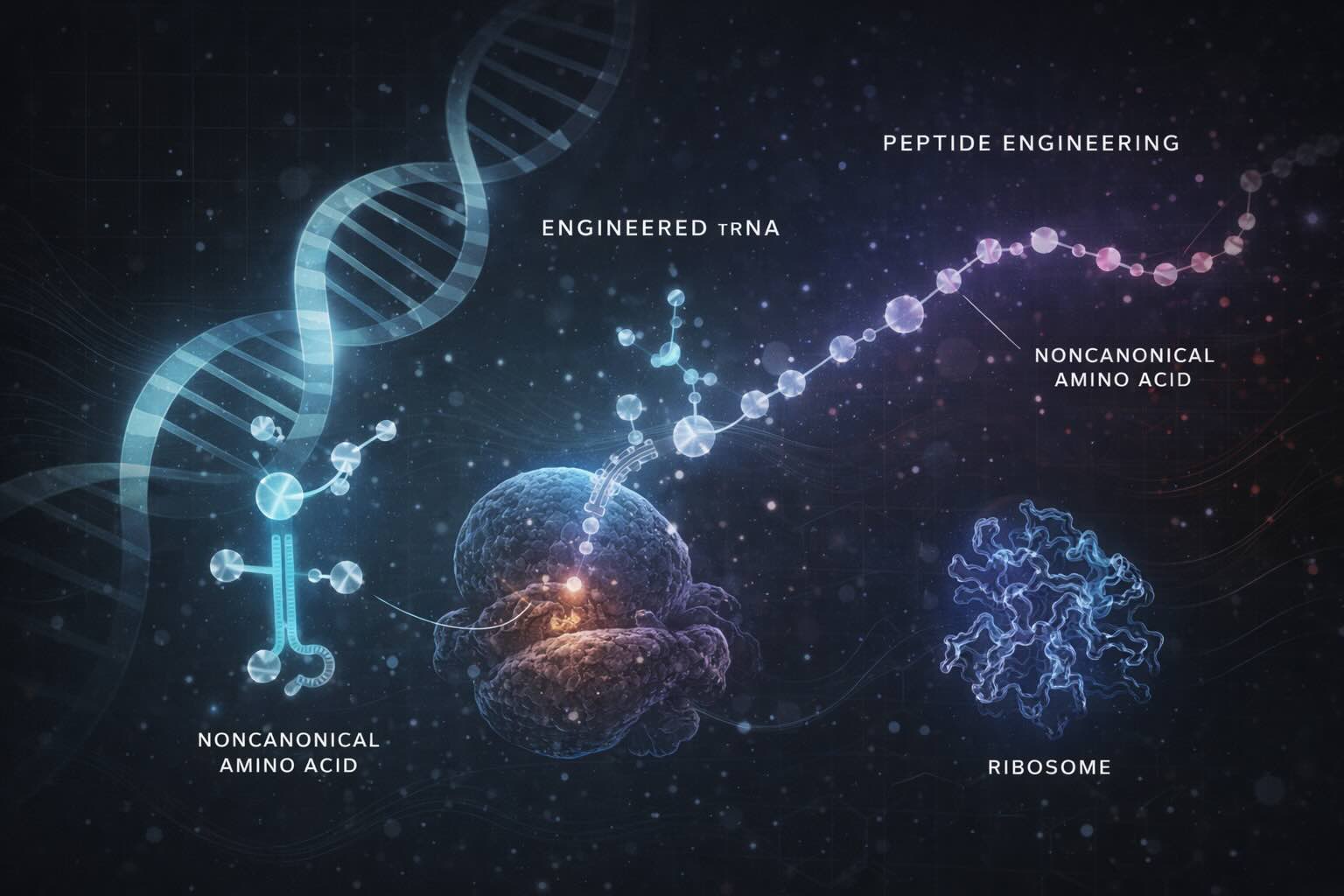
Peptides (short chains of amino acids) are increasingly important in medicine as highly specific and potent therapeutic agents. However, their natural structures are often vulnerable to rapid degradation and limited stability in the body. To address these challenges, researchers are exploring peptides that include chemically modified amino acids such as N-methyl-α-amino acids and d-α-amino acids, which provide enhanced stability and functional diversity. The ability to efficiently incorporate these unusual amino acids during peptide synthesis can revolutionize drug development, enabling the creation of novel therapeutics with improved efficacy. Recent advances in engineered transfer RNAs (tRNAs) are making this a reality by enhancing the ribosome’s capacity to include multiple exotic amino acids consecutively during protein synthesis.
Background: The Role of tRNAs in Peptide Synthesis
Proteins and peptides are synthesized by ribosomes, molecular machines that read messenger RNA (mRNA) and string together amino acids in the correct order using transfer RNAs (tRNAs). Each tRNA matches a specific amino acid to its corresponding mRNA codon through a specialized structure called the anticodon. This process uses the 20 canonical L-α-amino acids to build proteins, which naturally limits the chemical diversity of peptides.
Expanding peptide diversity by incorporating noncanonical or nonproteinogenic amino acids (npAAs) such as N-methyl-L-α-amino acids (MeAAs) and D-α-amino acids (D-AAs) can dramatically improve their pharmacological properties, including resistance to enzymatic degradation and enhanced membrane permeability. These amino acids, however, are poor substrates for the ribosome, mainly because natural tRNAs and the translation apparatus are optimized for the canonical amino acids.
To overcome this limitation, scientists have engineered tRNAs to improve the charging and incorporation of npAAs. For example, the specialized chimeric tRNAPro1E2, designed with optimized T-stem and D-arm structures, has demonstrated improved translation efficiency for MeAAs and D-AAs. However, incorporating multiple consecutive npAAs remained challenging due to diminished incorporation efficiency and peptidyl transfer limitations.
Key Findings: Engineering the Anticodon Stem of tRNAPro1E2
A recent study (PMID: 41732915) reports a groundbreaking improvement in this field by focusing on the anticodon stem region of the tRNAPro1E2. By screening 149 anticodon stem mutants, researchers identified variants that enhanced the ribosomal incorporation of N-methyl-L-leucine (MeLeu) and D-phenylalanine (D-Phe) at multiple consecutive codons, increasing efficiency by up to 4.5-fold.
Notably, 7 out of 11 effective variants showed altered mobility in gel electrophoresis experiments, suggesting that the mutations induced unique tRNA conformations favorable for peptidyl transfer reactions. Moreover, these engineered tRNAs also showed increased incorporation efficiencies for diverse MeAAs and D-AAs beyond the initial test amino acids, demonstrating broad applicability.
A key demonstration was the successful ribosomal synthesis of a macrocyclic peptide that contained three consecutive MeAAs and three consecutive D-AAs with high fidelity and quality — a milestone for peptide drug design and library construction incorporating multiple exotic amino acids.
Implications for Peptide Therapeutics and Synthetic Biology
These advances have significant implications for both therapeutic peptide development and synthetic biology. Incorporating multiple npAAs, especially in consecutive positions, enables the design of peptides with enhanced stability, conformational control, and biological activity. For example, N-methylation can improve resistance to proteases, increase membrane permeability, and modulate peptide secondary structures, making peptides more drug-like.
D-amino acids impart resistance against natural enzymatic degradation and can profoundly affect binding affinity and selectivity of peptides toward their targets. The ability to efficiently incorporate multiple consecutive D-AAs opens avenues for designing novel peptide scaffolds that were previously inaccessible by ribosomal synthesis.
Furthermore, engineered tRNAs capable of efficient multiple noncanonical amino acid incorporation can accelerate the construction of high-quality peptide libraries, which is invaluable for drug screening and discovery workflows. This technology complements other approaches such as cell-free translation systems, genetic code expansion, and chemical peptide synthesis, creating a versatile toolkit for peptide engineers.
Key Takeaways
- tRNAs are critical players in ribosomal peptide synthesis, and their structure influences amino acid incorporation efficiency.
- Engineering the anticodon stem of chimeric tRNAs can significantly improve the ribosomal incorporation of consecutive N-methyl and D-amino acids.
- Incorporation of multiple noncanonical amino acids in peptides enhances drug-like properties such as stability and bioavailability.
- These innovations enable the synthesis of complex macrocyclic peptides and diverse peptide libraries, crucial for next-generation therapeutics.
- Advances in tRNA engineering expand the chemical space accessible for ribosomal peptide synthesis.
Frequently Asked Questions
What are N-methyl-α-amino acids and why are they important?
N-methyl-α-amino acids are amino acids with a methyl group attached to the nitrogen atom of the peptide backbone. This modification improves peptide stability and membrane permeability, important for making peptides more drug-like.
Why are d-amino acids used in peptides?
D-amino acids are mirror images of the naturally occurring L-amino acids. Their incorporation can improve resistance to enzymatic degradation and influence peptide folding, enhancing therapeutic potential.
What role does tRNA play in peptide synthesis?
Transfer RNA (tRNA) carries specific amino acids to the ribosome and matches them to mRNA codons during translation, dictating the sequence of the synthesized peptide.
How does engineering the anticodon stem affect tRNA function?
Modifying the anticodon stem can alter tRNA structure and dynamics, enhancing the efficiency with which noncanonical amino acids are incorporated into growing peptides.
What are the potential applications of this technology?
It enables the development of novel peptide therapeutics with improved properties and facilitates synthetic biology approaches to create diverse peptide libraries for drug discovery.
Conclusion
Engineering the fundamental components of the translation machinery, such as tRNAs, represents a powerful frontier in expanding the chemical landscape of peptides. The ability to incorporate multiple consecutive noncanonical amino acids reliably opens exciting avenues in drug development, synthetic biology, and biomaterials. As these technologies mature, we can expect a generation of peptide-based therapeutics with enhanced stability, specificity, and novel functions, addressing a broad array of unmet medical needs.
[INTERNAL LINK: Peptide Therapeutics Overview]
For more detailed scientific insight, see the original study: PMID: 41732915.
References:
- Katoh T, Suga H. Engineering tRNAs for Noncanonical Amino Acid Incorporation. Nat Chem Biol. 2018;14(2):83-94.
- Gentilucci L, De Marco R, Cerisoli L. Chemical Modifications Designed to Improve Peptide Stability: Incorporation of Non-Natural Amino Acids, Pseudo-Peptide Bonds, and Cyclization. Curr Pharm Des. 2010;16(28):3185-203.
- Doi M, Yamaguchi S, Inokuma T, Suga H. Genetic Code Expansion in Cell-Free Translation Systems with Engineered tRNAs. ACS Chem Biol. 2020;15(9):2348-2356.
Related posts
Expanding the Horizon of Peptide Therapeutics: How Engineered tRNAs Enable Ribosomal Incorporation of Noncanonical Amino Acids
How Peptide-Based Therapies Like Exenatide Target Insulin Resistance and Inflammation in Type 2 Diabetes
BPC-157 and TB-500: Examining a Dual-Peptide Synergy in Tissue Repair Research
The Role of Tesamorelin in Modulating Growth Hormone Pathways and Metabolism
TB-500: Exploring the Synthetic Analog of Thymosin Beta-4 in Regenerative Research
Encoded in the Mitochondria: How MOTS-c Shapes Metabolic and Aging Pathways
A Three–Amino Acid Signal in Inflammation Control
IGF-1 LR3: Understanding the Long-Acting Insulin-Like Growth Factor in Research
Human Chorionic Gonadotropin 5000 IU: The Molecular Engine of Reproductive Science
Inside the Science of GLP-3: How Retatrutide Is Shaping Metabolic Innovation
The Glow Blend and the Biology of Peptide-Driven Repair
GHK-Cu and the Molecular Language of Tissue Repair
Recent Posts
- Expanding the Horizon of Peptide Therapeutics: How Engineered tRNAs Enable Ribosomal Incorporation of Noncanonical Amino Acids
- How Peptide-Based Therapies Like Exenatide Target Insulin Resistance and Inflammation in Type 2 Diabetes
- BPC-157 and TB-500: Examining a Dual-Peptide Synergy in Tissue Repair Research
- The Role of Tesamorelin in Modulating Growth Hormone Pathways and Metabolism
- TB-500: Exploring the Synthetic Analog of Thymosin Beta-4 in Regenerative Research